This course will examine the history of our planet, based upon scientific data, which shows very clearly that the age of the earth is about 4.5 billion years. We will not be looking specifically at many theological issues, however they will be addressed when necessary. First, let's examine one of the most frequently quoted evidences for a young earth, then we will move right on into earth's history.
Bishop Ussher and the Young Earth
Before the discoveries made by modern science, most people thought that the age of the earth was only about 6,000 years. This was based on estimates made by examining the first few chapters of the book of Genesis in the Bible. The most influential estimates from Genesis came from a Bishop in the Church of Ireland, James Ussher. He dated the creation to October 4004, B.C. This date was derived based upon the ages of people in the book of Genesis. It also assumes that there were no gaps in the genealogies of the Bible. We will not examine the genealogies here, but one very important point needs to be made. Since Ussher's dates are based upon the ages of the people in the Bible, Ussher IS NOT dating the age of the earth. He is dating the age of mankind, who was created at the very end of day six of creation.
Ussher's dates only go back to the end of the sixth day of creation. Since Ussher assumed that the days of creation were normal 24-hour days, he assumed that
mankind's age + 6 days = the age of the earth.
Since modern science has shown that the earth is billions of years old, we now know that Ussher's equation is wrong.
The Age of the Earth
The age of the Earth is around 4.54 billion years (4.54 × 109 years ± 1%). This age has been determined by radiometric age dating of meteorite material and is consistent with the ages of the oldest-known terrestrial and lunar samples. The Sun, in comparison, is about 4.57 billion years old, about 30 million years older.
Following the scientific revolution and the development of radiometric age dating, measurements of lead in uranium-rich minerals showed that some were in excess of a billion years old. The oldest such minerals analyzed to date – small crystals of zircon from the Jack Hills of Western Australia – are at least 4.404 billion years old. Comparing the mass and luminosity of the Sun to the multitudes of other stars, scientists state that the solar system cannot be much older than those rocks. Ca-Al-rich inclusions (inclusions rich in calcium and aluminium) – the oldest known solid constituents within meteorites that are formed within the solar system – are 4.567 billion years old, giving an age for the solar system and an upper limit for the age of Earth. It is hypothesised that the accretion of Earth began soon after the formation of the Ca-Al-rich inclusions and the meteorites. Because the exact accretion time of Earth is not yet known, and the predictions from different accretion models range from a few millions up to about 100 million years, the exact age of Earth is difficult to determine. It is also difficult to determine the exact age of the oldest rocks on Earth, exposed at the surface, as they are aggregates of minerals of possibly different ages. The Acasta Gneiss of Northern Canada may be the oldest known exposed crustal rock.
Development of Modern Geologic Concepts
Studies of strata, the layering of rock and earth, gave naturalists an appreciation that Earth may have been through many changes during its existence. These layers often contained fossilized remains of unknown creatures, leading some to interpret a progression of organisms from layer to layer.
Abū Rayhān Bīrūnī (11th century CE) discovered the existence of shells and fossils in regions that were once sea floor, but were later uplifted to become dry land, such as the Indian subcontinent. Based on this evidence, he realized that the Earth is constantly changing and proposed that the Earth had an age, but that its origin was too distant to measure. The principle of superposition of strata was first proposed by Avicenna (11th century). He outlined the principle while discussing the origins of mountains in The Book of Healing in 1027. Shen Kuo (11th century) also later recognized the concept of deep time.
Nicolas Steno (17th
century) was one of the first Western naturalists to appreciate the
connection between fossil remains and strata.[12]
His observations led him to formulate important
stratigraphic concepts (i.e., the "law
of superposition" and the "principle
of original horizontality").
In the 1790s, the British naturalist
William Smith
hypothesized that if two layers of rock at widely differing locations
contained similar fossils, then it was very plausible that the layers were
the same age. William Smith's nephew and
student,
John Phillips, later
calculated by such means that Earth was about 96 million years old.
The naturalist
Mikhail Lomonosov,
regarded as the founder of
Russian
science, suggested in the mid-18th century that Earth had been created
separately from the rest of the universe, several hundred thousand years
before. Lomonosov's ideas were mostly speculative, but in 1779, the
French
naturalist the
Comte du Buffon tried
to obtain a value for the age of Earth using an experiment: He created a
small globe that resembled Earth in composition and then measured its rate
of cooling. This led him to estimate that Earth was about 75,000 years old.
Other naturalists used these hypotheses to construct a history of Earth, though their timelines were inexact as they did not know how long it took to lay down stratigraphic layers. In 1830, the geologist Charles Lyell, developing ideas found in Scottish natural philosopher James Hutton, popularized the concept that the features of Earth were in perpetual change, eroding and reforming continuously, and the rate of this change was roughly constant. This was a challenge to the traditional view, which saw the history of Earth as static, with changes brought about by intermittent catastrophes. Many naturalists were influenced by Lyell to become "uniformitarians" who believed that changes were constant and uniform.
Early Calculations
 |
| William Thomson (Lord Kelvin) |
In 1862, the physicist William Thomson (who later became Lord Kelvin) of Glasgow published calculations that fixed the age of Earth at between 20 million and 400 million years. He assumed that Earth had formed as a completely molten object, and determined the amount of time it would take for the near-surface to cool to its present temperature. His calculations did not account for convection inside the Earth, which allows more heat to escape from the interior to warm rocks near the surface.
Geologists had trouble accepting such a short age for Earth. Biologists could accept that Earth might have a finite age, but even 100 million years seemed much too short to be plausible. Charles Darwin, who had studied Lyell's work, had proposed his theory of the evolution of organisms by natural selection, a process whose combination of random heritable variation and cumulative selection implies great expanses of time. (Geneticists have subsequently measured the rate of genetic divergence of species, using the molecular clock, to date the last universal ancestor of all living organisms no later than 3.5 to 3.8 billion years ago).
In a lecture in 1869, Darwin's great advocate, Thomas H. Huxley, attacked Thomson's calculations, suggesting they appeared precise in themselves but were based on faulty assumptions. The German physicist Hermann von Helmholtz (in 1856) and the Canadian astronomer Simon Newcomb (in 1892) contributed their own calculations of 22 and 18 million years respectively to the debate: they independently calculated the amount of time it would take for the Sun to condense down to its current diameter and brightness from the nebula of gas and dust from which it was born. Their values were consistent with Thomson's calculations. However, they assumed that the Sun was only glowing from the heat of its gravitational contraction. The process of solar nuclear fusion was not yet known to science.
Other scientists backed up Thomson's figures as well. Charles Darwin's son, the astronomer George H. Darwin of the University of Cambridge, proposed that Earth and Moon had broken apart in their early days when they were both molten. He calculated the amount of time it would have taken for tidal friction to give Earth its current 24-hour day. His value of 56 million years added additional evidence that Thomson was on the right track.
The last estimate Thomson gave, in 1897, was: "that it was more than 20 and less than 40 million year old, and probably much nearer 20 than 40".
In 1899 and 1900, John Joly of the Trinity College, Dublin calculated the rate at which the oceans should have accumulated salt from erosion processes, and determined that the oceans were about 80 to 100 million years old.
Radiometric Dating
Overview
Rock minerals naturally contain certain elements and not others. By the process of radioactive decay of radioactive isotopes occurring in a rock, exotic elements can be introduced over time. By measuring the concentration of the stable end product of the decay, coupled with knowledge of the half life and initial concentration of the decaying element, the age of the rock can be calculated. Typical radioactive end products are argon from potassium-40 and lead from uranium and thorium decay. If the rock becomes molten, as happens in Earth's mantle, such nonradioactive end products typically escape or are redistributed. Thus the age of the oldest terrestrial rock gives a minimum for the age of Earth assuming that a rock cannot have been in existence for longer than Earth itself.
Convective Mantle and Radioactivity
In 1892, Thomson had been made Lord Kelvin in appreciation of his many scientific accomplishments. Kelvin calculated the age of Earth by using thermal gradients, and arrived at an estimate of 100 million years old. He did not realize that Earth has a highly viscous fluid mantle, and this ruined his calculation. In 1895, John Perry produced an age of Earth estimate of 2 to 3 billions years old using a model of a convective mantle and thin crust. Kelvin stuck by his estimate of 100 million years, and later reduced the estimate to about 20 million years.
Radioactivity would introduce another factor in the calculation. In 1896, the French chemist A. Henri Becquerel discovered radioactivity. In 1898, Polish and French researchers, Marie and Pierre Curie, discovered the radioactive elements polonium and radium. In 1903 Pierre Curie and his associate Albert Laborde announced that radium produces enough heat to melt its own weight in ice in less than an hour.
Geologists quickly realized that the discovery of radioactivity upset the assumptions on which most calculations of the age of Earth were based. These calculations assumed that Earth and Sun had formed at some time in the past and had been steadily cooling since that time. Radioactivity provided a process that generated heat. George Darwin and Joly were the first to point this out, also in 1903.
Invention of Radiometric Dating
 |
| Ernest Rutherford |
Radioactivity, which had overthrown
the old calculations, yielded a bonus by providing a basis for new
calculations, in the form of
radiometric dating.
Ernest Rutherford and
Frederick Soddy,
working jointly at
McGill University, had
continued their work on radioactive materials and concluded that
radioactivity was due to a spontaneous transmutation of atomic elements. In
radioactive decay, an element breaks down into another, lighter element,
releasing alpha, beta, or gamma
radiation in the
process. They also determined that a particular isotope of a radioactive
element decays into another element at a distinctive rate. This rate is
given in terms of a "half-life",
or the amount of time it takes half of a mass of that radioactive material
to break down into its "decay product".
Some radioactive materials have short half-lives; some
have long half-lives.
Uranium and
thorium
have long half-lives, and so persist in Earth's crust, but radioactive
elements with short half-lives have generally disappeared. This suggested
that it might be possible to measure the age of Earth by determining the
relative proportions of radioactive materials in geological samples. In
reality, radioactive elements do not always decay into nonradioactive
("stable") elements directly, instead, decaying into other radioactive
elements that have their own half-lives and so on, until they reach a
stable element. Such "decay series", such as
the uranium-radium and thorium series, were known within a few years of the
discovery of radioactivity, and provided a basis for constructing techniques
of radiometric dating.
The
pioneers of radioactivity were
Bertram B. Boltwood, a
young chemist just out of
Yale, and the energetic Rutherford. Boltwood
had conducted studies of radioactive materials as a consultant, and when
Rutherford lectured at Yale in 1904, Boltwood
was inspired to describe the relationships between elements in various decay
series. Late in 1904, Rutherford took the first step toward radiometric
dating by suggesting that the
alpha particles released by radioactive
decay could be trapped in a rocky material as
helium
atoms. At the time, Rutherford was only guessing at the relationship between
alpha particles and helium atoms, but he would prove the connection four
years later.
Soddy and
Sir William Ramsay,
then at
University College in
London, had just determined the rate at which radium produces alpha
particles, and Rutherford proposed that he could determine the age of a rock
sample by measuring its concentration of helium. He dated a rock in his
possession to an age of 40 million years by this technique. Rutherford
wrote,
I came into the room,
which was half dark, and presently spotted Lord Kelvin in the audience and
realized that I was in trouble at the last part of my speech dealing with
the age of the earth, where my views conflicted with his. To my relief,
Kelvin fell fast asleep, but as I came to the important point, I saw the old
bird sit up, open an eye, and cock a baleful glance at me! Then a sudden
inspiration came, and I said, 'Lord Kelvin had limited the age of the earth,
provided no new source was discovered. That prophetic utterance refers to
what we are now considering tonight, radium!' Behold! the old boy beamed
upon me.
Rutherford assumed that the rate of decay of radium as determined by Ramsay and Soddy was accurate, and that helium did not escape from the sample over time. Rutherford's scheme was inaccurate, but it was a useful first step.
Boltwood focused on the end products of decay series. In 1905, he suggested that lead was the final stable product of the decay of radium. It was already known that radium was an intermediate product of the decay of uranium. Rutherford joined in, outlining a decay process in which radium emitted five alpha particles through various intermediate products to end up with lead, and speculated that the radium-lead decay chain could be used to date rock samples. Boltwood did the legwork, and by the end of 1905 had provided dates for 26 separate rock samples, ranging from 92 to 570 million years. He did not publish these results, which was fortunate because they were flawed by measurement errors and poor estimates of the half-life of radium. Boltwood refined his work and finally published the results in 1907.
Boltwood's paper pointed out that samples taken from comparable layers of strata had similar lead-to-uranium ratios, and that samples from older layers had a higher proportion of lead, except where there was evidence that lead had leached out of the sample. His studies were flawed by the fact that the decay series of thorium was not understood, which led to incorrect results for samples that contained both uranium and thorium. However, his calculations were far more accurate than any that had been performed to that time. Refinements in the technique would later give ages for Boltwood's 26 samples of 250 million to 1.3 billion years.
Arthur Holmes Establishes Radiometric Dating
Although Boltwood published his paper in a prominent geological journal, the geological community had little interest in radioactivity. Boltwood gave up work on radiometric dating and went on to investigate other decay series. Rutherford remained mildly curious about the issue of the age of Earth but did little work on it.
Robert Strutt tinkered
with Rutherford's helium method until 1910 and then ceased. However,
Strutt's student
Arthur Holmes became interested in
radiometric dating and continued to work on it after everyone else had given
up. Holmes focused on lead dating, because he regarded the helium method as
unpromising. He performed measurements on rock samples and concluded in 1911
that the oldest (a sample from Ceylon) was about 1.6 billion years old.
These calculations were not particularly trustworthy. For example, he
assumed that the samples had contained only uranium and no lead when they
were formed.
More important,
in 1913 research was published showing that elements generally exist in
multiple variants with different masses, or "isotopes".
In the 1930s, isotopes would be shown to have nuclei with differing numbers
of the neutral particles known as "neutrons".
In that same year, other research was published establishing the rules for
radioactive decay, allowing more precise identification of decay series.
Many geologists felt these new discoveries made radiometric dating so complicated as to be worthless. Holmes felt that they gave him tools to improve his techniques, and he plodded ahead with his research, publishing before and after the First World War. His work was generally ignored until the 1920s, though in 1917 Joseph Barrell, a professor of geology at Yale, redrew geological history as it was understood at the time to conform to Holmes's findings in radiometric dating. Barrell's research determined that the layers of strata had not all been laid down at the same rate, and so current rates of geological change could not be used to provide accurate timelines of the history of Earth.
Holmes's persistence finally began to pay off in 1921, when the speakers at the yearly meeting of the British Association for the Advancement of Science came to a rough consensus that Earth was a few billion years old, and that radiometric dating was credible. Holmes published The Age of the Earth, an Introduction to Geological Ideas in 1927 in which he presented a range of 1.6 to 3.0 billion years. No great push to embrace radiometric dating followed, however, and the die-hards in the geological community stubbornly resisted. They had never cared for attempts by physicists to intrude in their domain, and had successfully ignored them so far. The growing weight of evidence finally tilted the balance in 1931, when the National Research Council of the US National Academy of Sciences finally decided to resolve the question of the age of Earth by appointing a committee to investigate. Holmes, being one of the few people on Earth who was trained in radiometric dating techniques, was a committee member, and in fact wrote most of the final report.
The report concluded that radioactive dating was the only reliable means of pinning down geological time scales. Questions of bias were deflected by the great and exacting detail of the report. It described the methods used, the care with which measurements were made, and their error bars and limitations.
Modern Radiometric Dating
Radiometric dating continues to be the predominant way scientists date geologic timescales. Techniques for radioactive dating have been tested and fine-tuned for the past 50+ years. Forty or so different dating techniques are utilized to date a wide variety of materials, and dates for the same sample using these techniques are in very close agreement on the age of the material.
Possible contamination problems do exist, but they have been studied and dealt with by careful investigation, leading to sample preparation procedures being minimized to limit the chance of contamination. Hundreds to thousands of measurements are done daily with excellent precision and accurate results. Even so, research continues to refine and improve radiometric dating to this day.
Why Meteorites Were Used
Today's accepted age of Earth of 4.54 billion years was determined by C.C. Patterson using uranium-lead isotope dating (specifically lead-lead dating) on several meteorites including the Canyon Diablo meteorite and published in 1956.
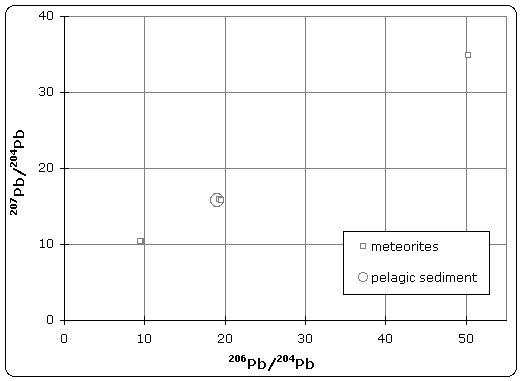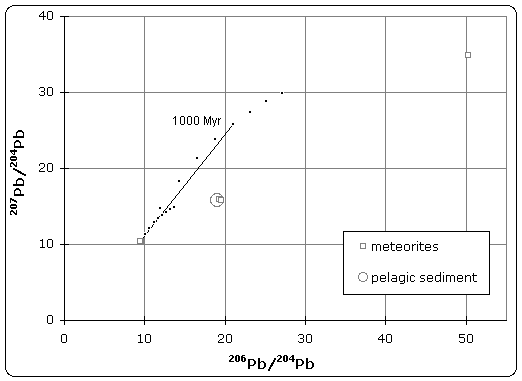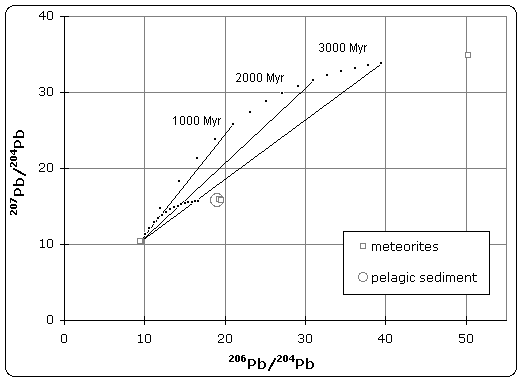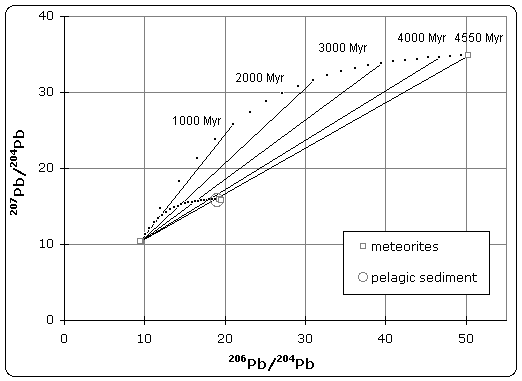 |
| Lead isotope isochron diagram showing data used by Patterson to determine the age of the Earth in 1956. (Click to enlarge) |
The quoted age of Earth is derived, in part, from the Canyon Diablo meteorite for several important reasons and is built upon a modern understanding of cosmochemistry built up over decades of research.
Most geological samples from Earth
are unable to give a direct date of the formation of Earth from the solar
nebula because Earth has undergone differentiation into the core, mantle,
and crust, and this has then undergone a long history of mixing and unmixing
of these sample reservoirs by
plate tectonics,
weathering and
hydrothermal circulation.
All of these processes may adversely affect isotopic
dating mechanisms because the sample cannot always be assumed to have
remained as a closed system, by which it is meant that either the parent or
daughter
nuclide (a species of
atom characterised by the number of neutrons and protons an atom contains)
or an intermediate daughter nuclide may have been partially removed from the
sample, which will skew the resulting isotopic date. To mitigate this effect
it is usual to date several minerals in the same sample, to provide an
isochron. Alternately, more than one dating
system may be used on a sample to check the date.
Some meteorites are furthermore considered to represent
the primitive material from which the accreting solar disk was formed.
Some have behaved as closed systems (for some isotopic systems) soon after
the solar disk and the planets formed. To date, these assumptions are
supported by much scientific observation and repeated isotopic dates, and it
is certainly a more robust hypothesis than that which assumes a terrestrial
rock has retained its original composition.
Nevertheless, ancient
Archaean lead
ores
of
galena have been used
to date the formation of Earth as these represent the earliest formed
lead-only minerals on the planet and record the earliest homogeneous
lead-lead isotope systems on the planet. These have returned age dates of
4.54 billion years with a precision of as little as 1% margin for error.
Statistics for several meteorites that have undergone
isochron dating are as follows:
-
St. Severin (ordinary chondrite)
-
Pb-Pb isochron - 4.543 +/- 0.019 GY
-
Sm-Nd isochron - 4.55 +/- 0.33 GY
-
Rb-Sr isochron - 4.51 +/- 0.15 GY
-
d. Re-Os isochron - 4.68 +/- 0.15 GY
-
-
Juvinas (basaltic achondrite)
-
Pb-Pb isochron ..... 4.556 +/- 0.012 GY
-
Pb-Pb isochron ..... 4.540 +/- 0.001 GY
-
Sm-Nd isochron ..... 4.56 +/- 0.08 GY
-
Rb-Sr isochron ..... 4.50 +/- 0.07 GY
-
-
Allende (carbonaceous chondrite)
-
Pb-Pb isochron ..... 4.553 +/- 0.004 GY
-
Ar-Ar age spectrum ..... 4.52 +/- 0.02 GY
-
Ar-Ar age spectrum ..... 4.55 +/- 0.03 GY
-
Ar-Ar age spectrum ..... 4.56 +/- 0.05 GY
-
Why the Canyon Diablo Meteorite was Used
 |
| Fragment of the Canyon Diablo Meteorite (Picture Source) |
The Canyon Diablo meteorite was used because it is a very large representative of a particularly rare type of meteorite that contains sulfide minerals (particularly troilite, FeS), metallic nickel-iron alloys, plus silicate minerals. This is important because the presence of the three mineral phases allows investigation of isotopic dates using samples that provide a great separation in concentrations between parent and daughter nuclides. This is particularly true of uranium and lead. Lead is strongly chalcophilic and is found in the sulfide at a much greater concentration than in the silicate, versus uranium. Because of this segregation in the parent and daughter nuclides during the formation of the meteorite, this allowed a much more precise date of the formation of the solar disk and hence the planets than ever before.
The Canyon Diablo date has been backed up by hundreds of other dates, from both terrestrial samples and other meteorites. The meteorite samples, however, show a spread from 4.53 to 4.58 billion years ago. This is interpreted as the duration of formation of the solar nebula and its collapse into the solar disk to form the Sun and the planets. This 50 million year time span allows for accretion of the planets from the original solar dust and meteorites.
The moon, as another extraterrestrial body that has not undergone plate tectonics and that has no atmosphere, provides quite precise age dates from the samples returned from the Apollo missions. Rocks returned from the moon have been dated at a maximum of around 4.4 and 4.5 billion years old. Martian meteorites that have landed upon Earth have also been dated to around 4.5 billion years old by lead-lead dating. Lunar samples, since they have not been disturbed by weathering, plate tectonics or material moved by organisms, can also provide dating by direct electron microscope examination of cosmic ray tracks. The accumulation of dislocations generated by high energy cosmic ray particle impacts provides another confirmation of the isotopic dates. Cosmic ray dating is only useful on material that has not been melted, since melting erases the crystalline structure of the material, and wipes away the tracks left by the particles.
Altogether, the concordance of age dates of both the earliest terrestrial lead reservoirs and all other reservoirs within the solar system found to date are used to support the hypothesis that Earth and the rest of the solar system formed at around 4.53 to 4.58 billion years ago.
Helioseismic Verification
The radiometric date of meteorites
can be verified with studies of the Sun. The Sun can be dated using
helioseismic methods
that strongly agree with the radiometric dates found for the oldest
meteorites.
End of Lesson
Additional Reading (Optional): Radiometric Dating: A Christian Perspective
Return to the Old Earth Ministries Online Earth History Curriculum homepage.
Source: Age of the Earth